Quality of life and mental health in children with long COVID
Study design, participants and procedure
This study employed an observational, cross-sectional, multi-cohort design to evaluate HRQoL and mental and social health of children (8-18 years old) with PLC, by comparing them to previously published data of children with other chronic health conditions (CHC) and children from the general population (GP) during the pandemic.
Pediatric Long COVID (PLC) cohort
The PLC cohort consisted of children who participated in the Post COVID Syndrome (POCOS)-study, a prospective, observational cohort study conducted at the Amsterdam University Medical Center (Amsterdam UMC), a tertiary care hospital based in Amsterdam, the Netherlands. The POCOS-study aimed to describe clinical characteristics, investigate underlying mechanisms, and identify biomarkers for PLC25. Participants consisted of children with physician-diagnosed Long COVID, according to the WHO definition, which defines Long COVID as a condition of newly onset or persisting symptoms at least three months after SARS-CoV-2 infection, lasting for at least 2 months with no other explanation19. Participants were included between May 2021 and March 2023. The Medical Ethics Committee of the Amsterdam UMC, location AMC, approved the study (METc 2021_126). All participants and/or caregivers provided informed consent. The PLC cohort consisted of 106 children, with an age range of 8 to 18 years old, of which 31% were male.
Reference data
Chronic health conditions (CHC) cohort
The CHC cohort consisted of children with chronic somatic diseases treated at the Emma Children’s Hospital, Amsterdam UMC. Participants were selected from those already undergoing patient-reported outcome measures (PROMs) as part of their standard care, and invited to complete additional questionnaires in early May, 2020, to evaluate mental and social health during the first COVID-19 lockdown. All children and parents provided informed consent and the study was approved by the Medical Ethics Committee of the Amsterdam UMC. The CHC cohort consisted of 90 children, with an age range of 8 to 18 years old, of which 56% were male18. Data used for this study was obtained through collaboration with the Department of Child and Adolescent Psychiatry and Psychosocial Care of the Amsterdam UMC. More details on the CHC cohort prior to the pandemic can be found in Table 2 of the publication by van Muilekom et al.26.
General population (GP) cohort
Data from Dutch children during the COVID-19 pandemic were collected through an independent online research agency, “Panel Inzicht,” between April 10 and May 5, 2020, to evaluate mental and social health during the first COVID-19 lockdown. The sample was representative of the Dutch general population within 2.5% on most key demographics (age, gender, ethnicity, region, and educational level). Additionally, as a reference group, mean scores from the same sample of Dutch children before the pandemic (2018) have been added, but no statistical tests were conducted with this data. All children and parents provided informed consent and the studies were approved by the Medical Ethics Committee of the Amsterdam UMC. The GP cohort consisted of 844 children, with an age range of 8 to 18 years old, of which 47% were male1. Data used for this study was obtained through collaboration with the Department of Child and Adolescent Psychiatry and Psychosocial Care of the Amsterdam UMC.
Data collection and outcome measures
Data from all cohorts were primarily collected via online questionnaires administered through the research website of the KLIK Patient-Reported Outcome Measures (PROM) portal27. The Pediatric Quality of Life Inventory 4.0 (PedsQL 4.0) PROM was completed by the PLC- and CHC-cohorts, and the Patient-Reported Outcomes Measurement Information System (PROMIS) measures assessing anxiety, anger, depressive symptoms, sleep-related impairment, and peer relationships, were completed by all three cohorts. For children from the PLC-cohort, because PROMs were collected for standard clinical care, patients received questionnaires before their outpatient visit, and results were discussed during the visit.
Pediatric Quality of Life Inventory 4.0 (PedsQL 4.0)
Domains and scoring
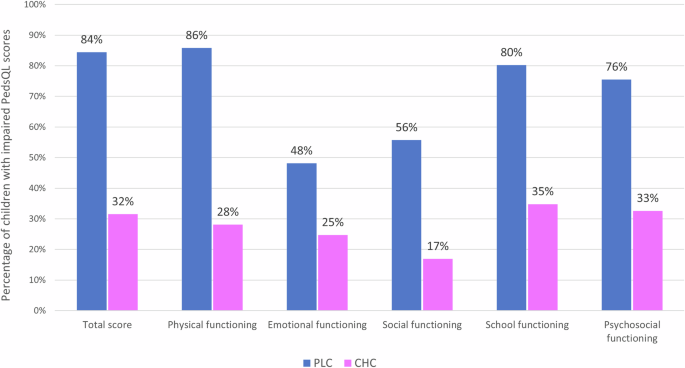
The Dutch PedsQL 4.0 is a comprehensive PROM comprising 23 items intended to evaluate the self-reported HRQoL of children. The PedsQL is available in versions for certain age categories, of which two have been used in this study: 8-12 years old and 13-18 years old. It encompasses four domains of HRQoL: Physical Health (8 items), Emotional Functioning (5 items), Social Functioning (5 items), and School Functioning (5 items). Employing a recall period of one week, respondents rated items on a scale from 1 (Never a problem) to 5 (Almost always a problem). These responses were then linearly transformed to a 0–100 scale (0 = 100, 1 = 75, 2 = 50, 3 = 25, 4 = 0), with higher scores indicating better functioning and better HRQoL. The Total PedsQL score was calculated as the mean of all items across the entire questionnaire, also ranging from 0 to 100. The Psychosocial Functioning PedsQL score was calculated as the mean of the Emotional, Social and School scores. Validation studies have confirmed the suitability of the Dutch PedsQL for clinical use in the Netherlands28.
Severity cutoffs
Cut-off points for at-risk status of impaired HRQoL by the PedsQL 4.0 were set at 1 standard deviation (SD) below the population mean29. The population mean and SD used to create categories of Normal and Impaired HRQoL for this study were based on psychometric properties of PedsQL 4.0 in the Dutch pediatric population28.
Patient-Reported Outcomes Measurement Information System (PROMIS) questionnaires
Domains and scoring
PROMIS item banks and scales, developed and validated using modern psychometric techniques, measure generic unidimensional domains across physical, social, or mental health. These measures were administered as Computerized Adaptive Tests (CAT) format, in which subsequent questions are chosen based on the answers given earlier, resulting in reliable scores with a minimal number of items. Five Dutch PROMIS pediatric measures, including Anxiety, Anger, Depressive Symptoms, Sleep-related Impairment (SRI), and Peer Relationships, were completed by all children and adolescents aged 8 to 18 years. Two additional Dutch PROMIS pediatric measures, Fatigue and Mobility, were completed by the PLC cohort. These measures, recognized by the American Psychiatric Association (APA) as level-2 assessment tools for psychiatric disorder monitoring from DSM-V, utilize a 7-day recall period and are scored on a five-point Likert scale. Total scores were derived by transforming item scores into T-scores ranging from 0 to 100. For PROMIS Anxiety, Anger, Depressive Symptoms, Sleep-related Impairment, and Fatigue, higher scores indicate more symptoms and/or worse functioning. For PROMIS Peer Relationships and Mobility, lower scores indicate more symptoms and/or worse functioning.
Severity cutoffs
Severity cutoffs were determined per PROMIS domain based on psychometric properties of PROMIS questionnaires in Dutch populations30,31,32,33. For domains where higher scores reflect poorer functioning, severity groups were defined as Minimal ( <75th percentile), Moderate (75–94th percentile), and Severe ( ≥95th percentile). For domains where higher scores reflect greater functioning, severity cutoffs were defined as Good ( >25th percentile), Fair (6th–25th percentiles), and Poor ( ≤5th percentile).
Statistics and reproducibility
IBM Statistical Package for Social Sciences (SPSS) version 28.0 was used to perform statistical analyses. Descriptive analyses were performed to summarize participant characteristics and mean with standard deviation (SD) were calculated for PedsQL and PROMIS T-scores for all three cohorts.
A one-way analysis of covariance (ANCOVA) was conducted to compare the mean PedsQL scores between the PLC- and CHC-cohorts, and the mean PROMIS T-scores between the PLC-, CHC-, and GP-cohorts whilst controlling for age and sex. Levene’s test and normality checks were carried out and the assumptions met. Chi-squared tests were conducted to compare severity categories between cohorts and the relative risk (RR) with 95% confidence intervals (95% CI) were reported. The RR represents the risk of a child with PLC having a severe score compared to a child with a CHC or a child from the GP-cohort. A ratio higher than 1 indicated more risk. Bonferroni-corrected post-hoc t-tests were performed to determine which cohorts differed significantly (defined as a p-value < 0.05) from each other.
Comparative analyses for PROMIS T-scores between the CHC- and GP-cohort have been previously published18 and will therefore not be performed in the current study. Each participant was considered a biological replicate, and no technical replicates were used. All statistical tests were two-sided and reproducibility of the analyses was ensured through use of predefined protocols and consistent data processing across cohorts.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
link